11th Week: The s,r and p-Process: Difference between revisions
| Line 10: | Line 10: | ||
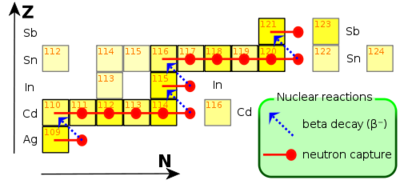
[[Image:S-process-elem-Ag-to-Sb. | [[Image:S-process-elem-Ag-to-Sb.png|thumb|right|400 px|The S-process acting in the range from Ag to Sb (Image is taken from the main wikipedia).]] | ||
The S-process or slow-neutron-capture-process is a nucleosynthesis process that occurs at relatively low neutron density and intermediate temperature conditions in stars. Under these conditions the rate of neutron capture by atomic nuclei is slow relative to the rate of radioactive beta-minus decay. A stable isotope captures another neutron; but a radioactive isotope decays to its stable daughter before the next neutron is captured. This process produces stable isotopes by moving along the valley of beta stability in the chart of isotopes. The S-process produces approximately half of the isotopes of the elements heavier than iron, and therefore plays an important role in the galactic chemical evolution. | The S-process or slow-neutron-capture-process is a nucleosynthesis process that occurs at relatively low neutron density and intermediate temperature conditions in stars. Under these conditions the rate of neutron capture by atomic nuclei is slow relative to the rate of radioactive beta-minus decay. A stable isotope captures another neutron; but a radioactive isotope decays to its stable daughter before the next neutron is captured. This process produces stable isotopes by moving along the valley of beta stability in the chart of isotopes. The S-process produces approximately half of the isotopes of the elements heavier than iron, and therefore plays an important role in the galactic chemical evolution. | ||
Revision as of 09:36, 24 April 2009
Production of elements heavier than iron
As the nuclear charge increases, the transmission probability decreases drastically. This in turn makes the charged cross sections to be very small and therefore at moderate stellar temperatures it is very difficult to form heavy elements. However, the elements heavier than are observed which requires explanation. Below we explain the main possible processes that might have occured in stellar nucleosynthesis.
Neutron capture
Neutron capture plays an important role in the cosmic nucleosynthesis of heavy elements. In stars, it can proceed in two ways - as a rapid process (an r-process) or a slow process (an s-process). By neutron capture, nuclei of masses greater than 56 can be formed that could not be formed by thermonuclear reactions, i.e., by nuclear fusion.
The S-process
The S-process or slow-neutron-capture-process is a nucleosynthesis process that occurs at relatively low neutron density and intermediate temperature conditions in stars. Under these conditions the rate of neutron capture by atomic nuclei is slow relative to the rate of radioactive beta-minus decay. A stable isotope captures another neutron; but a radioactive isotope decays to its stable daughter before the next neutron is captured. This process produces stable isotopes by moving along the valley of beta stability in the chart of isotopes. The S-process produces approximately half of the isotopes of the elements heavier than iron, and therefore plays an important role in the galactic chemical evolution.
The S-process is believed to occur mostly in Asymptotic Giant Branch stars. In contrast to the R-process which is believed to occur over time scales of seconds in explosive environments, the S-process is believed to occur over time scales of thousands of years. The extent to which the s-process moves up the elements in the chart of isotopes to higher mass numbers is essentially determined by the degree to which the star in question is able to produce neutrons, and by the amount of iron in the star's initial abundance distribution. Iron is the "starting material" for this neutron capture - beta-minus decay sequence of synthesizing new elements.
The main neutron source reactions are: