PHZ3400-09 Atoms: Difference between revisions
| Line 2: | Line 2: | ||
==Van der Waals Force== | ==Van der Waals Force== | ||
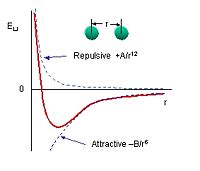
The Van der Waals Force, also known as the London Force, arises when two | The Van der Waals Force, also known as the London Force, arises when two inert atoms bond. Inert atoms have filled electron shells, are very stable, and only affected a small amount when bonding to another inert atom. They act as if they are solid spheres. The bonding of the atoms is highly dependent upon the distance that separates them and can be described by the Lennard-Jones potential. When the atoms are separated by a large distance, the atoms are attracted because they induce an electric dipole moment on each other and become polarized. When the atoms are too close together, the repulse each other because of the Pauli Principle which does not allow two electrons to occupy the same quantum state. | ||
[[Image:lennardjones.JPEG|thumb|left|200px|]] | [[Image:lennardjones.JPEG|thumb|left|200px|]] | ||
Revision as of 16:34, 23 January 2009
Please add pictures, as apporpriate, with captions, through cut-and-past from PDF files or from other Web downloads.
Van der Waals Force
The Van der Waals Force, also known as the London Force, arises when two inert atoms bond. Inert atoms have filled electron shells, are very stable, and only affected a small amount when bonding to another inert atom. They act as if they are solid spheres. The bonding of the atoms is highly dependent upon the distance that separates them and can be described by the Lennard-Jones potential. When the atoms are separated by a large distance, the atoms are attracted because they induce an electric dipole moment on each other and become polarized. When the atoms are too close together, the repulse each other because of the Pauli Principle which does not allow two electrons to occupy the same quantum state.
Ionic Bond
Examples?
Covalent Bond
It would be nice to add here a discussion of various SP hybrids leading to different bonding schemes for carbon. If possible, a separate page devoted to organic molecules or even polymers, based on the above.