PHZ3400-09 Atoms: Difference between revisions
| Line 2: | Line 2: | ||
==Van der Waals Force== | ==Van der Waals Force== | ||
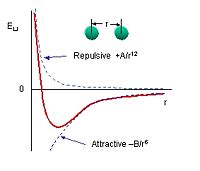
[[Image:lennardjones.JPEG|thumb|right|200px|Lennard-Jones potential. The force between atoms is the negative derivative of this curve.]] | [[Image:lennardjones.JPEG|thumb|right|200px|Lennard-Jones potential. The force between atoms is the negative derivative of this curve.]] | ||
The Van der Waals Force, also known as the London Force, arises when two inert atoms bond. Inert atoms have filled electron shells, are very stable, and only affected a small amount when bonding to another inert atom. They act as if they are solid spheres. The bonding of the atoms is highly dependent upon the distance that separates them and can be described by the Lennard-Jones potential. When the atoms are separated by a large distance, the atoms are attracted because they induce an electric dipole moment on each other and become polarized. When the atoms are too close together, the repulse each other because of the Pauli Principle which does not allow two electrons to occupy the same quantum state. | |||
==Atomic Bonding: Chemistry Recap== | ==Atomic Bonding: Chemistry Recap== | ||
Revision as of 19:30, 25 January 2009
Please add pictures, as apporpriate, with captions, through cut-and-past from PDF files or from other Web downloads.
Van der Waals Force
The Van der Waals Force, also known as the London Force, arises when two inert atoms bond. Inert atoms have filled electron shells, are very stable, and only affected a small amount when bonding to another inert atom. They act as if they are solid spheres. The bonding of the atoms is highly dependent upon the distance that separates them and can be described by the Lennard-Jones potential. When the atoms are separated by a large distance, the atoms are attracted because they induce an electric dipole moment on each other and become polarized. When the atoms are too close together, the repulse each other because of the Pauli Principle which does not allow two electrons to occupy the same quantum state.
Atomic Bonding: Chemistry Recap
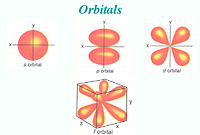
From chemistry we should remember that electrons form orbitals around the nuclei of atoms. The electrons form their shells in a specific order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p and so on.
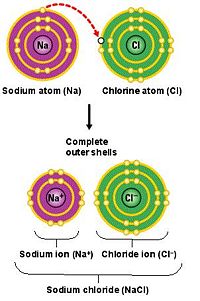
Ionic Bond
Ionic bonding is characterized by a greedy atom who steals and electron from another atom. For example, a Cl atom, with one 3s electron outside a filled shell, steals an electron from a Na atom, which lacks an electron for a filled 3p shell, forming Cl- and Na+. The energy lost in this exchange is recovered by moving the ions close together in order to increase their Coulomb potential forming NaCl. If the ions move too close together, their electron will overlap forcing Na's electrons in the 2p shell to move up to the 3p shell or higher to accommodate and this takes energy. This energy might be thought of as a potential of repulsion and increases as the separation between the ions decreases.
Covalent Bond
The atoms involved in a covalent bond share electrons. Ideally, the electrons are shared equally, but the atoms must be identical for this to be true. One instance of this is the hydrogen molecule. Most often, the electrons will reside nearer to one of the nuclei. An example of this is in water. Since the oxygen molecule has a much greater positive charge, the electrons tend to stay closer to the oxygen than to the hydrogens creating an electric dipole. For example, in the semiconductor GaAs, the shared electrons are located nearer to As than Ga. This illustrates the state in which some compound form bonds that are not purely covalent nor ionic, but in some state between. The bond between GaAs is 66% covalent.
- It would be nice to add here a discussion of various SP hybrids leading to different bonding schemes for carbon. If possible, a separate page devoted to organic molecules or even polymers, based on the above.
Metallic Bond
It is better to use the name metallic bonding becuase there is no single bond that characterizes this. Metallic bonding is a a collective "glue" of delocalized electrons that hold the positive ions togeather. Delocalization arises because the zero point energy of an electron is very large, the electron is not confined to a fixed position and it vibrates with a large amplitude These collective electrons is sometimes referred to as the "sea of electrons" through which electricity is conducted. The structure of materials that form metallic bonding is mainly determined on how the positive ions are arranged.
Hydrogen Bond
Hydrogen bonding occurs between two molecules when a somewhat positive hydrogen region of a molecule is electrostatics attracted to a negatively charged region of a another molecule and forms a weak bond. The bond strength is about 20 times less than that of a covalent bond. The simplest example of a hydrogen bond is between water molecules. Another example is hydrogen bonding between different parts of a long DNA molecule to form a helical shape.