4th Week: Decays, Tunneling and Cross Sections
Nuclear Radiation
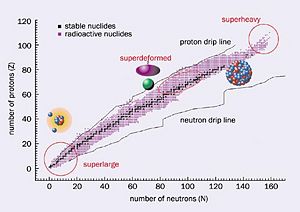
When looking at the table of nuclides it is obvious that the valley of stability is only a small percentage of all known nuclei. So most nuclei are considered unstable. To become stable these nuclides emit ionizing particles and radiation, resulting in a loss of energy from the original nuclei. This Process is called radioactive decay.
Alpha Decay and Fission
Alpha decay is the transformation of a parent nucleus to a daughter nucleus through the emission of a helium nucleus. This decay is usually seen in heavier elements with too many protons. This causes excessive repulsion due to the coulomb force. In an attempt to get rid of this excessive repulsion the nucleus emits an alpha particle.
Alpha decay and nuclear fission are both considered to be a two body disintegration. In the case of a alpha decay, the daughter nucleus is at rest and most of the released energy is carried away by the alpha particle. For Fission the energy is distributed between the two daughter nuclei.
Beta Decay
There are three types of Beta decays, -decay, -decay, and electron capture. All three of these decays can be characterized by and .
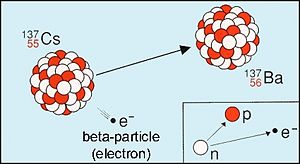
The first one, decay, occurs when a nucleus with an over abundance of neutrons transforms to a more stable nucleus by emitting an electron. This process can be denoted as:
From electric-charge conservation, the proton number in such decays is increased by one unit.
Likewise, a -decay occurs when a proton-rich nucleus emits a positron, therefore reducing the nuclear charge by one.
The final case, electron capture, is similar to -decay because it is another way for a proton -rich nucleus can reduce its nuclear charge by the capture of an electron.
Gamma Decay