PHZ3400-09 Emergence in Condensed Matter
I want you to type, and type, and type here... add as much information as comes to mind... formulas, links... whatever helps make the story more convincing... This is the Age of Wikipedia, after all...
Do we have to watch every atom?
The answer to this depends on the context of the problem. If one is looking at the motion of a soccer ball the answer is no. The particles of the air inside the ball can move randomly. These particles can appear to move erratically. However, if one were to look at the overall motion, the center of mass motion, of the aggregate, the motion of the group is greatly simplified. On the other hand, if one wanted to know the air pressure inside the ball, knowing properties of the individual particles is probably more convenient.
If one wants to know if one should look at every atom, one should decide based on whether the property desired is macroscopic or microscopic.
Defining Macroscopic Bodies
A macroscopic body is made up of many microscopic bodies usually on the order of at least one mole. The amount of particles in one mole is given by Avagadro's number, on the order of . Were every particle to be watched, there would be something on the order of states to solve for. A significantly high number capable of being too much for even the best computers.
Rather than doing that, most of the time only the center of mass is needed to draw a complete picture about the object. The Center of Mass is found using the following expression:
Many phases of matter
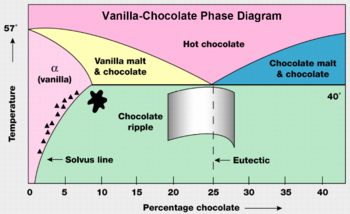
Matter comes in many forms macroscopically. Water comes in solid (like the snowflake shown), liquid, and gaseous forms. What governs these transitions for water is temperature. To compound this phenomenon further, compounds, such as carbon chains, can have different types of the same state, which for solid carbon includes graphite, coal, and diamonds. These different forms of the same state are known as allotropes. A pressure-temperature graph would show that these different types of solid carbon are formed at different pressures. Another method of phase shifting can be seen in the vanilla-chocolate phase diagram. As one can see the percent of chocolate in the vanilla-chocolate mixture also can cause a phase shift. Though this example appears simplistic, as compounds do exhibit property changes as the proportions of the constituent elements change.
How important is Quantum mechanics?
The cause of these phase changes is an interesting question. Can these phase changes be found by classical or quantum mechanical means?
For example, why don't we suffer the fate of Rumpelstinskin (drop through the floor)? This is because objects are solid. But is solidity a quantum or classical phenomena? Solidity is most likely a macroscopic property as solids contain many particles. But classical physics gives no reason for objects to be solid other than molecular vibrations from the temperature of the solid. So ideally one could manipulate the temperature and affect the solidity of the floor. Except for the extreme case where the temperature is brought to the level where the floor dissociates, the temperature does not affect solidity. However, quantum mechanics can solve this problem. Heisenberg's uncertainty principle forces the electrons of the floor to be discrete values. This also means that the orbitals are incompressible unless the electrons can orbit in the same energy level (that is, possess non distinct quantum numbers). Pauli's exclusion principle and Fermi-Dirac statistics show that this is also not a possibility, because electrons are fermions and therefore cannot occupy the same quantum state simultaneously. So despite solidity being macroscopically observed, it is quantum mechanics that keeps solids from passing through each other.