3rd Week: Equation of State & Basic Properties of Nuclei C
Nuclei
Constituents
Nuclei are made up of two fermions (collectively called nucleons), a neutrally charged Neutron of mass 939 MeV and the positively charged Proton of mass 938 MeV. They are held in atomic nuclei despite their electrical repulsion by the Strong Nuclear Force. As the range of this force is only on the order of a femtometre, nuclei are only several fm large.
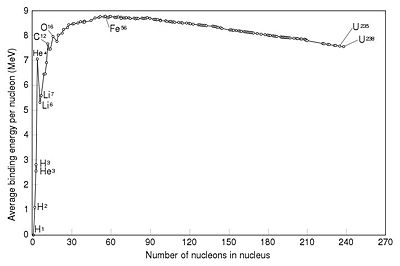
The strength of the Coulomb force between charged particles at this range is very large, and so the energy needed to bind them together is also very large, indeed it is detectable as a mass difference between bound and unbound nucleons. By measuring this mass difference for each nuclei, and dividing by the number of nucleons in each nucleus, we can get a measure of how tightly each nucleon is bound.
From the graph we can see that light nuclei (below Fe) become more tightly bound with increasing nucleon number, while beyond iron nuclei are less tightly bound. This makes sense when we consider that the strong force only acts at a distance comparable to the size of a single nucleon, in large nuclei an extra proton will only see the repulsive force of distant nucleons, not their attraction. Also beyond the region of very light nuclei, the energy per nucleon is nearly flat, at least to a rough estimate.
There are several models of nuclei to reproduce these binding energies:
Nuclei Models
Independent Particle Model
Droplet Model
Shell Model
The Shell Model of the nucleus is a model of the atomic nucleus which uses the Pauli principle to describe the structure of the nucleus in terms of energy levels. The Shell Model accounts for many features of these nuclear energy levels. According to this model, the motion of each nucleon is governed by the average attractive force of all the other nucleons. The resulting orbits form "shells," just as the orbits of electrons in atoms do. As nucleons are added to the nucleus, they drop into the lowest-energy shells permitted by the Pauli Principle which requires that each nucleon have a unique set of quantum numbers to describe its motion. When a shell is filled with maximum number of protons or neutrons allowed, the nucleus becomes extremely stable, and if the shell is filled with the maximum number of both protons and neutrons, it is even more stable still. The numbers of nucleons which fill these shells to the maximum allowed according the the Pauli Exclusion Principle are referred to as "magic numbers", and if a shell is filled with the max protons and neutrons, the nucleus is said to be "doubly magic". The first 6 magic numbers are 2, 8, 20, 28, 50, 82, and 126.