Crystal growth
Crystal Growth is a major stage of crystallization that occurs after nucleation. It involves the addition of new atoms, ions and other materials to the crystal base to form the lattice of the crystal.
Nucleation
Nucleation is the first stage in the crystallization process. It is a localized physical process that involves the budding of lattice points. Nucleation is a slow process because in order for it to start, the atoms or ions have to be able to become attracted to one another. Nucleation in the case of crystals occurs when there is a supersaturated system and the sites start to cluster on the nanometer scale. These sites when stable become the nuclei or the start of the crystal. The sites have a higher concentration of the solution that is being used to make the crystal, and only once they are stable will they start to be nuclei. However, if the nuclei sites are unstable, they will redissolve back into the solution to try again later. The nuclei need to be at a critical size in order for nucleation to continue over to the crystal growth stage. There are two ways that a solution can go through nucleation: homogeneous nucleation and heterogeneous nucleation.
Nucleation isn't only a process for crystallization; it also occurs elsewhere in nature. Clouds use nucleation to form water droplets. Carbon dioxide bubbles nucleate when you release the pressure on the container holding them. The most visible effect of nucleation happens when you introduce Mentos to Diet Coke.
Homogeneous Nucleation
Homogeneous nucleation occurs when the solution is made up entirely of a uniform content. An example of this is purified water. This typically takes the longest as purified solutions have a lower energy state that they need to reach in order to nucleate. This energy state is lower than their normal ground state energy and it has to be drained off in order for the nucleation to happen. Thus it requires a large amount of energy to get started.
Examples of Homogeneous Nucleation
- Purified Water freezes at -40 degrees Celsius
- Perfect Diamonds have no impurities
Heterogeneous Nucleation
Heterogeneous nucleation happens when there are impurities in the solution that is being used for crystallization. These impurities can be naturally in there or artificially put in, but either way, they form the center for a nucleation point. The impurities create a site where surface energy is lower and easier to overcome for the case of nucleation sites.
Heterogeneous nucleation is the most common form of nucleation in nature. Water forms ice crystals when there is a speck of dust to nucleate around, and others nucleate as well.
The most common form of nucleation used for the artificial growth of crystals is this point where some sort of impurity is introduced into the solution to act as something for the crystals to grow off of. This can be as simple as a piece of string, or as complex as scratches in the container holding the crystallization solution.
Examples of Heterogeneous Nucleation
- Normal water freezes at 0 degrees Celsius. This is due to dust particles being there to nucleate around.
- Important in condensation of rain droplets
- Causes Snowflakes
- Causes Hail
Crystal Growth
Crystal growth occurs when the amount of nuclei that have reached the critical size start to gain even more mass. As more sites become available, and as long as supersaturation occurs, the crystal will grow larger and retain its lattice shape. Each nucleation site is a lattice point, and the crystallization will occur around it. Once the lattice sites are uniform, the crystal begins to take its macroscopic shape. Depending on the conditions of the growing area, nucleation could dominate the crystal growth or vice versa. This results in different crystal shapes for even crystals made of the same elements.
Examples of Natural Crystals
- Ice
- Honey
- Stalagmites and Stalactites
- Gem stones
Gem Stones
Gem stones are naturally occuring crystals that grow under high heat and pressure. They are often found underground where the ambient pressure is high and there is a good amount of heat. Diamond, a gem stone formed from carbon atoms can only become this gem stone when under these conditions. Otherwise, the carbon atoms will become a different lattice shape that forms something like graphite or coal.
Crystal Growth Techniques
The ability to control the shape of the crystal and its properties depends on the techniques used during the growth and the amount of nucleation sites prepared for the solution. Many techniques for crystal growth exist, and each of them has a reason that it is used. The techniques listed in this article are used to artificially create crystals for various reasons. How effective the techniques are, the crystals that can be produced from them, and why the techniques are used will be explained in each section.
Floating Zone Image Furnace Growth
A technique known as floating zone image furnace growth is used at Florida State University to produce crystalline solids using various non-organic elements. This is a non-traditional crystal growth technique that allows for an odd way for the crystals to form.
How to do the Technique.
1. Figure out the materials: In order for the crystal to be grown, the grower has to pen the chemical reaction that he or she wants to have happen. Using the chemical formula reaction, and depending on the size of the crystal desired, the grower will then figure out the exact mass of each element that is required in order for the crystal to form properly in the reaction. If there is too much or too little of the elements, the reaction will not happen.
2. Prepare the materials: This is where the grower will measure out the powdered forms of each element. Often, due to the fact that the materials are stored in contact with atmosphere, the materials are hydrated. This means that they have bonded with the hydrogen atoms in the atmosphere. These have to be heated up so that they can become purified elements. The process that heats them up is long, almost eighteen hours long.
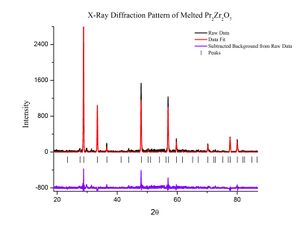
3. Test the materials: The materials are tested using an x-ray diffraction technique to determine whether they reacted properly. If it is found that the materials have not reacted properly, the materials must be prepared over again.
4. Create the rods.: If it is found that the materials react the way that they need to react, then they are molded into a rod and cooked so that they will stay in that rod formation. This is another long process that takes close to eighteen hours. The rod is then split in half for the next step.
5. Cook the rods in the image furnace: The image furnace is an oven powered by a special light bulb that has a tungsten filament. There are two concave gold mirrors that focus the light on one spot such that it can increase its heat to a high temperature. The two rod pieces are then positioned vertically, one above the other. The rods are rotated and the furnace is turned on. The heat produced by the furnace causes the upper rod to melt and then fall in droplets onto the bottom rod.
6. Allow the rods to cool: The rods have to cool slowly. This allows the crystal's lattice to form as the amorphous solid cools. The reason for the slow cooling is that there is a domain wall that has to be overcome at the high temperature in order for the crystal to form. The domain wall shifts as the crystal slowly cools. If speed cooling is used, the lattice domain wall will freeze in place, unable to allow the crystal to form.
7. Check the finished crystal: Once again x-ray diffraction is used to determine whether or not the crystal has successfully become what it is that the grower wanted. If the diffraction is of the appropriate type, the structure of the crystal can be determined to be what was wanted or not.
Quartz Hydrothermal Growth
Quartz is a crystal that has a very high melting point, 1670 degrees Celsius. It also has a high chemical resistance. This makes forming crystals by melting or flux techniques difficult. Cooling through the Curie Point results in twin crystals, thus in order to make a single crystal, a different technique must be applied. The process in question is a solution growth process known as hydrothermal growth, and this is one of the few methods that can result in a single crystal.
Technique Description
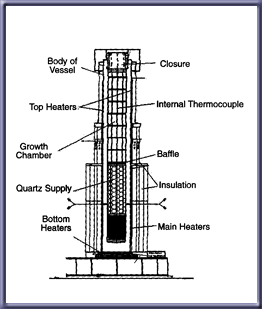
Hydrothermal growth is done inside of a set of steel pressure vessels known as called autoclaves. A nutrient material supply is put in the bottom of the autoclave, and the top of the autoclave is covered with seed crystals. A metal plate with at least one hole cut in it to provide a constant opening is placed between the two halves of the autoclave, and then an aqueous solution, which acts as a mineralizer and is often an alkali material, is placed in some fixed volume of the autoclave.
After the materials are added in, the autoclave is sealed. Upon sealing, the autoclave is heated using external heaters to temperatures between 300 and 400 degrees Celsius. The internal pressure of the autoclave is controlled by the temperature and the contents of the autoclave itself.
Ideally, there should be a difference in temperature between the two halves such that the lower half is at a higher temperature than the upper half. The lower half at this point is called the dissolving half, while the upper half is the growing half. Due to the temperature gradient that develops in this process, a natural convection current forms in the autoclave. The dense fluid at the bottom rises to encounter the cooler temperatures of the growing half which shall be further referred to as the growing chamber. Upon reaching the cooler temperatures of the growing chamber, the liquid creates a supersaturated solution. The crystal base from the supply precipitates on the seed crystals, causing the crystal to grow. The now cooler dense liquid falls down and the process begins over again. This process continues until the crystal is formed to the specified size.
This still does not guarantee that the crystal will be a single crystal rather than a crystalline solid. Further tests are needed to determine this.
Single Crystal
A single crystal is a crystal where the lattice is completely aligned throughout the entirety of the crystal. This is a difficult thing to accomplish, and there is no guarantee that the process that the grower is going through will produce a single crystal.
The crystal growth process is not perfect due to entropic variations at even the nanoscopic level. These variations can prevent even the most perfect setting from producing a single crystal. Single Crystal growth has been described as the “black magic” of physics due to the fact that it is something that either happens or does not. A grower that can consistently create single crystals of significant size would be looked upon as a valuable resource.
References
1. Pawlicki, Alison, Personal Interview, 16 April 2009
2. "Crystallization" CUBoulder Organic Chemistry Lab, http://orgchem.colorado.edu/hndbksupport/cryst/cryst.html 4/17/09
3. "Quartz Hydrothermal Growth" Sawyer Research Products, http://www.roditi.com/SingleCrystal/Quartz/Hydrothermal_Growth.html 4/17/09