Liquid Crystal
Liquid Crystal is a phase of matter all its own that has characteristics similar to that of solids and liquids, but possesses unique properties that has made it a large area of research for the past 100 years and shows promise of more innovation in today's environmentally conscious society.
History
Many scientists had already observed this phenomenon of liquid crystals prior to its "discovery", however the Austrian botanist Friedrich Reinitzer is more often then not given credit for its discovery. While experimenting with cholesterol benzoate in 1888, Reinitzer noted that it the substance had two melting points. The first melting point occurred at 145.5°C, where it melted to a cloudy liquid phase, and 178.5°C, where it melted to a clear liquid phase. He also observed some strange color phenomena as the temperature changed.
Reinitzer then sent some of his samples to a German physicist, Otto Lehmann, who had a state of the art polarized microscopic, which he outfitted with heating stage so he could precisely control the temperature of his specimens. Lehmann first described Reinitzer's sample as floating crystals and crystalline fluids, but after more experimentation became convinced that the cloudy liquid was a uniform phase all its own. This led him to coin the term liquid crystal to describe this new phase of matter.
In the early 1900s, many advances were made in the understanding liquid crystals. In 1922, French physicist, Georges Freidel, gave a detailed description of different liquid crystal phases, coining many terms that are still used today. Freidel also explored the orienting effect of an electric field on liquid crystals.
After World War II interest in liquid crystals began to wane. Many scientists believed that all of the important problems regarding liquid crystals had been solved. However, in the 1960s scientists began to re-examine liquid crystals and continued the progress of the pre-war era. It was discovered that liquid crystalline substances were extremely sensitive to very small changes in temperature. Researchers at RCA found that with an electrical voltage applied, a thin layer of liquid crystal was capable of switching from cloudy to clear.[1]
Overview
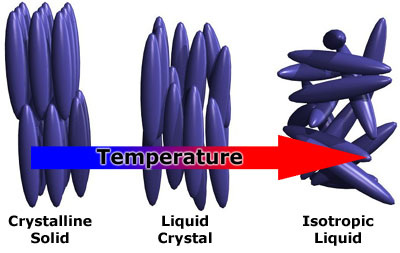
Solids possess both positional order, where molecules are constrained to occupy only certain positions, and orientaitional order, where each molecule is oriented with respect to one another. When a material transitions to a liquid both the positional and orientational order are lost, and the material becomes isotropic, or uniform in all directions. In the liquid crystal phase, molecules, or mesogen, display a wide range of positional and orientational orders, leading to several subphases. These subphases are defined by the amount of order present in the liquid crystal.[2]
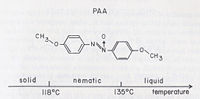
Liquid crystals are a phase of matter between the solid phase and the liquid phase. If you have a material that has a liquid crystal phase you would observe the two melting points that Reinitzer observed, one from the solid to liquid crystal transition and the second from the liquid crystal to isotropic liquid transition.
However, this is only the case in thermotropic liquid crystal, where phase transitions are dependant on the temperature. It is possible for liquid crystal phase to be dependant on the concentration of component in another, this is called lyotropic liquid crystal.[3]
Some molecules are more likely to posses a liquid crystal phase than others. There are 3 properties that make a molecule a good candidate to become liquid crystal[4]:
(1) Elongated in shape: elongated molecules have stronger attractive forces when they are aligned parallel to one another.
(2) Rigid in the center:
(3) Flexible on the ends: allows for easier positioning in between other molecules
The rod shaped liquid crystal is the most common type, however there are others. In 1977, disclike molecules were discovered that formed a liquid crystal phase. This type of liquid crystal is known as discotic.
Since the liquid crystal phase is between that of solid and liquid, it displays characteristics common to both, as well as some unique characteristic all its own. Molecules are free to move about as in a liquid, however they on average spend more time pointing along the direction of orientation. Liquid crystal will also flow and take the shape of its container, however it also display a cloudiness which suggests that it is different from the liquid phase. Many unique characteristics of liquid crystals stem from the fact that it is an anisotropic phase. In other words, any property measured along one direction of space will have different values when measured along any other direction.
Methods to Measure Orientational Order
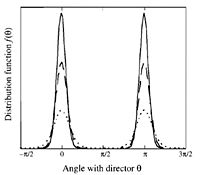
To quantitatively describe the amount of orientational order present in a subphase of liquid crystal the average direction of the mesogen, is measured with respect to a direction of orientation known as the director. Each molecule makes an angle, with respect to the director. This angle is then measured and averaged over all molecules.
The more orientational order present the closer the average will be to zero. An alternative way to do this is to take the average angle of one molecule over a certain time, instead of looking at different molecule. However, this only works under the assumption that all molecules undergo random motion. Thankfully, this assumption has never been contradicted experimentally.
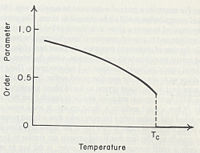
To create a more standard method of measuring orientational order we introduce the order parameter . Where once again we take the angle with respect to the director, however this time we use the function,
Now integrating over the solid angle,
where is the fraction of molecules in a solid angle which are oriented at an angle of to the director.[5]
Now perfect oriental orientation will have an average of 1 and no orientational order will have an average of 0.
Subphases
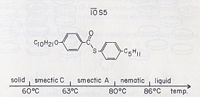
Although many compounds exhibit only one liquid crystal phase, it is not unusual for a single substance to posses more than one.[6] To the right are phase diagrams of two different molecules that form into different subphases at different temperatures.
Nematic
Nematic Phse can be thought of as a liquid that has long range orientational order, but lacks postional order.[7]
Smectic
Molecules in the smectic phase have orientational order, as in the nematic phase. In addition, they gain long-range positional order in one dimension. Essentially the molecules align themselves in sheets (1D positional order), however within these sheets there is only orientational order.[8]
Smectic phases are broken into many sub-classes designated with a letter(i.e. smectic A, smectic B, etc). There are many sub-classes that range from having one dimensional positional order to positional order in three dimensions, as well as varying orientational orders. Two of the most common sub-classes are the smectic A and smectic C sub-classes. In the smectic A phase the director is aligned perpendicular to the planes.[9]
While in the smectic C phase the director is aligned at some angle other than 90° to the planes.[10]
Chirality
Both the nematic and smectic phase can exist in alternate forms named, chiral nematic and chiral smectic respectively. The word chiral simply means twisted. So in each one of these cases the direction of the director changes with position, creating a helical pattern. This phase is present in molecules that favor bonding at slight angles with one another.[11]
Influences of an External Field
One of the reasons liquid crystal has so many applications is because of its interactions with an external field, be it electrical or magnetic. Solids, liquids, and gases respond to electric and magnetic fields, but the response is minimal even when strong fields are applied. Liquid Crystal, on the contrary, respond to even weak electric and magnetic fields with significant structural change.[12]
Electric Field
Liquid crystal is usually composed of neutral atoms, however in some the bonding between each molecule causes one part of the molecule to be slightly more positive on one end and slightly more negative on the other (here we are considering elongated mesogen). This causes a permanent electric dipole within the liquid crystal.
Now when an electric field is applied to the liquid crystal the molecule will rotate until it is aligned with the electric field. This means, depending on the bonding and the distribution of charge within the molecule, that it will tend to align either parallel to the applied electric field or perpendicular to it.
If this is not the case and the bonding does not form a separation of charges, an applied electric field can still induce an electric dipole. In this case the electric field causes a force and displaces some atomic charged particles within the molecule. An induced electric dipole is usually much weaker than a permanent electric dipole, however it still produces the same effect and orients itself along the electric field.
Now to display liquid crystal's anisotropic properties, consider a layer of liquid crystal in between two plates, such that the director is stuck in place. Now lets apply an electric field to the liquid crystal at various angles to the director. If we were to do this we would notice that each direction we apply the field at we will observe a different degree of polarization. To quantify this we introduce electric susceptibility, , which is defined as the ratio of polarization, , to the strength of the applied electric field, .
The chart to the right considers a case where the charges are separated along the long axis of the molecule and as you can see much more polarization can be induced when the applied field is in the same direction as the director.
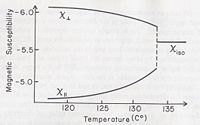
Magnetic Field
An applied magnetic field on liquid crystal produces very similar results to that of an electric field. Although, liquid crystal atoms do not act as a permanent magnetic dipole, a magnetic field will induce a magnetic dipole. This means that liquid crystal forming molecules are diamagnetic. In the presence of a magnetic field the molecule will then tend to orient itself either parallel or perpendicular to the applied field.
Again we can display the anisotropic properties of liquid crystal, by introducing the magnetic susceptibility, , which we define as as the ratio of magnetization, , to the strength of the applied magnetic field, .
Applications
Liquid crystal technology provides for a wide range of application, but the place it has made the most impact on is the display industry. LCD televisions, having gained broad-based consumer popularity in the past 15-20 years, dominate the television market today. The fourth quarter of 2007 provided an important milestone in the industry when LCD television sales surpassed cathode ray tube televisions for the first time.[15]
Even though, we are exposed to liquid crystal technology in displays most often, does not mean the technology is limited to this industry. Some liquid crystal materials change color when stressed or heated and therefore are used to measure stress distributions and heat flow in materials, as well as novelty items such as mood rings.[16]
Displays
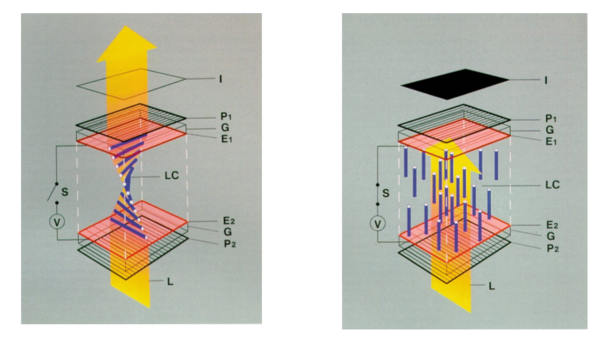
In a simple Liquid Crystal Display (i.e. no color), there are two polarizers placed a small distance apart from each other. The polarizers are set up, so that their angles of polarization are perpendicular to each other. This means if light were to pass from the first straight through to the second then an observer would not see any light through the polarizer.
Now a thin layer of liquid crystal is placed in between the polarizers. With the liquid crystal in between the polarizers an observer would notice that light is now showing through the polarizers. This is because the liquid crystal is anisotropic and essentially acts as a polarizer itself and polarizes the light at some intermediate angle between that of the two surrounding polarizers. Now we can apply a force field (electric, magnetic, etc...) to the liquid crystal to align the particles in a direction that lets the light pass through unaffected. This means that again the observer will see no light.
So using this method we are able to insert electrodes into the area of the liquid crystals to create a localized electric field that will block out any light in that area, creating a black image on the observer's screen.
Liquid crystal displays, such as televisions and computer monitors, use the same process to create the images, however color filters are also used. Here each picture element, or pixel, consists of a red, green, and blue color filter. Different colors are then synthesized by varying the brightness of light through each one of these filters.
Smart Glass
Another application of liquid crystals, which is sure to gain popularity in this environmentally conscious time we live in is smart glass. A smart glass is intended to have the ability to control the amount of light, and heat, passing through. With the push of a button, the glass can change from transparent to completely opaque. Unlike blinds, smart windows are capable of partially blocking light while maintaining a clear view of what lies behind the window.[17] Not only do smart windows control the amount of light that enters a room, but by making the window opaque, you can control the amount of heat that enters from the sun.
This technology takes advantage of the unique ability for liquid crystal to align itself in the presence of an electric field. The liquid crystal is sandwiched in the center of the window with transparent electrodes attached on both sides. When no voltage is supplied to the electrodes the liquid crystal orients itself randomly , thereby scattering any light passing through the window. When a small voltage is applied the electrodes create an electric field, which align the molecules of liquid crystal. This creates a minimum of scattering and the light passes through unaffected.
The smart in smart glass comes from the fact that these windows are being outfitted with artificial intelligence to control the environment in a room without user interaction. It is the hope that this technology will help to create smart buildings, which minimize the impact on the environment by conserving energy.
As far as the actual impact of smart windows, a recent EPA funded report found that "...the SW [Smart Window] will save 23% percent of the energy that would otherwise go towards room lighting..." and estimates that the initial $1,000 cost of the window would pay for itself in less than three years.[18]
References
[9,10]Chandrasekhar, Liquid Crystals, 1977
Chigrinov, Liquid Crystal Devices: Physics and Applications, 1999
[1,3,4,6,11,12,13]Collings, Liquid Crystals, 1990
[5,14]Jones, Soft Condensed Matter, 2002
[7,8]Kumar, Liquid Crystals, 2001
[2]Saeva, Liquid Crystals: The Fourth State of Matter, 1979
[15] Wikipedia Article, Liquid Crystal Display
[16] Wikipedia Article, Liquid Crystal
[17] Glass On Web, Electronic Smart Glasses
[18] EPA Final Report, Smart Windows for Smart Buildings
External Links
- Liquid Crystal Institute at Kent State University
- Wikipedia Article, Liquid Crystal
- Wikipedia Article, Liquid Crystal Display
- Glass On Web, Electronic Smart Glasses
- Wikipedia Article, Smart Glass
- EPA Final Report, Smart Windows for Smart Buildings
- ↑ Collings, Liquid Crystal
- ↑ Saeva, Liquid Crystal, The Fourth State of Matter
- ↑ Collings, Liquid Crystal
- ↑ Collings, Liquid Cryrals
- ↑ Jones, Soft Condensed Matter
- ↑ Collings, Liquid Crystals
- ↑ Kumar, Liquid Crystals
- ↑ Kumar, Liquid Crystals
- ↑ Chandrasekhar, Liquid Crystals
- ↑ Chandrasekhar, Liquid Crystals
- ↑ Collings, Liquid Crystal
- ↑ Collings, Liquid Crystal
- ↑ Collings, Liquid Crystals
- ↑ Jones Soft Condensed Matter
- ↑ http://en.wikipedia.org/wiki/Liquid_Crystal_Display
- ↑ http://en.wikipedia.org/wiki/Liquid_Crystal
- ↑ http://www.glassonweb.com/articles/article/192
- ↑ http://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/7172/report/F