Metallic Glass
A metallic glass, also known as an amorphous metal or glassy metal, is a metallic material that lacks a crystalline structure. Metals typically have a highly-ordered crystalline structure, whereas a metallic glass is amorphous in nature, much like a glass. Various methods are used to fix the metal's structure in an amorphous state, and the result is a material which possesses a number of novel properties with promising future applications.
How it's made
All metallic glasses are produced by cooling a metal alloy quickly enough so that crystals do not have time to form. Just how this is achieved has evolved since these materials were first discovered.
Rapid cooling
The first metallic glass was produced in 1959 by W. Klement (Jr.), Willens and Pol Duwez at Caltech. It was a 75/25 gold/silicon alloy that was solidified by cooling it from the liquid state at an extremely high rate of roughly one million Kelvin per second. This rapid cooling process forced the material into an amorphous state by not allowing the metal atoms enough time to order themselves into a crystal lattice. An unfortunate limitation of this process was that metallic glasses could only be formed as extremely thin samples, generally under a hundred microns thick, as heat needed to be removed quickly in order to frustrate crystallization of the material. This rapid cooling technique remained the only viable method for producing metallic glasses for nearly three decades, and with the limitations of the method, few applications could be realized. The use of melt spinning techniques to rapidly cool ribbons of the material allowed for some improvement in production, but the fundamental thickness limitation held back advancement in the field for quite some time.
Although the production method didn't much change, the necessary rate of cooling did see a dramatic decrease over the years. In 1969, a 77.5/16.5/6 palladium/silicon/copper alloy was developed that could be cooled at a much slower rate on the order of several hundred Kelvin per second. Various other alloy formulations allowed continued progress toward still lower minimum cooling rates.
Crystal frustration
It wasn't until the early 1990s that theoretical advances led to a significant reduction in cooling rates. Building on the theories of Harvard researcher David Turnbull, by 1990 Akahisa Inoue's group at Japan's Tohoku University had succeeded in producing samples of metallic glass up to a quarter of an inch thick. Dubbed bulk metallic glasses for their hefty achievable thickness, these revolutionary materials were made possible by composing the alloy from three or more elements whose atomic radii differ by at least twelve percent. These alloys solidify via something known as the "confusion principle", whereby the the significant variation in atomic sizes frustrates the formation of a crystal lattice, thus "confusing" the atoms as to what crystalline structure to settle into. This slows down the crystallization process to such a high degree that such alloys can be made to settle into an amorphous structure at a much slower rate of cooling than traditional metallic glasses, thus allowing much greater thicknesses to be achieved.
In 1992, William Johnson's group at Caltech developed the first of a series of metallic glass alloys known as Vitreloy, the first of which consisted of a 41.2/22.5/13.8/12.5/10 zirconium/beryllium/titanium/copper/nickel blend. This material cooled so slowly that it could be cast to thicknesses of over an inch.
Structure
The structure of metallic glass is markedly different than that of an ordinary metal.
Ordinary metal
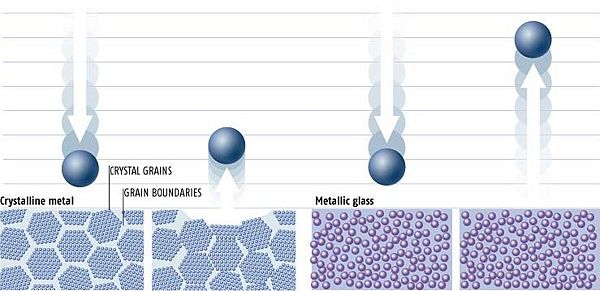
Typical metals have a highly-ordered crystalline structure consisting of repeating unit cells. An important consequence of this regular structure is that crystals have cleavage planes, so forces applied along these planes can lead much easier to deformation of the crystal. Metals are also frequently polycrystalline, meaning that they are not composed of a single crystal, but rather multiple crystals with different orientations. As an ordinary metal cools, crystallization begins randomly at different locations within the material, producing numerous individual crystals known as crystallites. As these crystallites continue to grow, eventually they run up against other crystallites. The surfaces along which these growing crystals collide are known as grain boundaries, and these are an important defect present in metals and many other crystalline materials. The presence of grain boundaries in a metal leads to a number of generally undesirable effects, including decreased electrical and thermal conductivity and reduced strength, as well as facilitating the onset of corrosion.
Metallic glass
In contrast, metallic glasses have an amorphous structure, and thus they have no cleavage planes or grain boundaries. This leads to increased strength and resistance to wear and corrosion, among other desirable properties, as compared to ordinary metals. The atoms in a sample of a modern metallic glass vary greatly in size, which frustrates the crystallization process and, coupled with rapid cooling from the liquid state, results in a random arrangement of atoms rather than an ordered crystal.
Properties
The amorphous structure of metallic glass endows it with many novel properties.
Elasticity
Because metallic glasses lack grain boundaries, they possess much greater elasticity to metallic glass than seen in ordinary metal. When a force impacts a piece of ordinary metal, the metal deforms and grain boundaries shift permanently, thus absorbing some of the energy of the blow. Deformations in a piece of metallic glass, on the other hand, are nearly reversible, as the lack of grain boundaries means that the atoms are actually overall more tightly packed than in an ordinary metal, and so there is much less available space to allow for permanent deformations. The result is that all of the energy of a blow to a piece of metallic glass is returned to the striking object.
{{#ev:youtube|nOHoRdgx4uA}}
Strength
The lack of grain boundaries also means that metallic glasses are less susceptible to the typical defect-related failures of ordinary metals. Metallic glasses tend to have higher tensile strength, for example that of Vitreloy is nearly twice that of high-grade titanium. There are limitations, however, as this increased tensile strength comes at a cost of greatly reduced ductility.
Injection moldability
Perhaps one of the most intriguing properties of metallic glasses from a manufacturing standpoint is that because are true glasses they can be injection molded like plastic. Ordinary metals must be worked after solidification in order to reduce grain boundaries and other defects and harden the crystal structure, as well as additional processing such as buffing to smooth out surface defects and give a mirror finish. Metallic glasses require no such post-processing, and injection-molded metallic glass parts emerge shiny and ready to use, possessing all of their desired properties from the start. What's more, unlike ordinary crystalline metals, metallic glasses exhibit very little volume change upon solidification, making casting even more straightforward.
Additional properties
Metallic glasses possess other unique properties. Among these is that particular metallic glass alloys, in particular those between the glass-forming elements boron, silicon, or phosphorus with magnetic elements such as iron, cobalt, or nickel, are magnetic materials possessing low coercivity (the strength of an applied field required to demagnetize a material after its magnetization has been saturated) and high electrical resistance. This leads to low losses to eddy currents when the material is in an alternating magnetic field, making them useful in the magnetic cores of transformers.
Stress and fatigue
Like all new materials, metallic glasses are not without obstacles to be overcome. Their main disadvantage as a structural material is that, like other glasses, they are highly susceptible to fatigue, which can lead to sudden catastrophic failure.
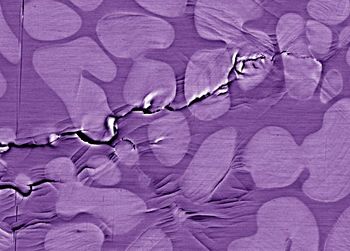
The problem
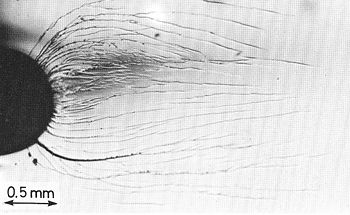
If a crack develops in a crystalline material such as an ordinary metal, its growth is soon interrupted when the energy dissipates along grain boundaries between the crystals. When a crack develops in a piece of glass, however, it propagates relatively unimpeded within its amorphous structure. Being true glasses, metallic glasses suffer from the same problem. The irony is that one of the main features that make metallic glasses so promising as a material of the future -- namely, their ability to deform reversibly and thus absorb little energy from a strike -- gives them little ability to stop cracks from propagating all the way through their structure.
The solution
Researchers at the U.S. Department of Energy’s Lawrence Berkeley National Laboratory and the University of California at Berkeley, working with colleagues at the California Institute of Technology, have solved the fracture problem by developing a method to allow a second, crystalline phase of metal to grow in thin, treelike structures throughout the bulk metallic glass. These crystalline inclusions absorb the energy of propagating cracks, giving the resulting composite material good fatigue resistance while still retaining the novel properties of an amorphous metal.
Uses
Because of their many novel properties, metallic glasses have many exciting potential uses, a number of which are already a reality.
Today
Due to their unique magnetic properties (as mentioned above), particular amorphous metal alloys have been in use in transformer cores since the early 1980s. This was the first commercial use of metallic glass, and the devices have led to a marked increase in transformer efficiency since their widespread adoption in the United States.
The increased wear resistance of metallic glasses over ordinary metals has led to a growing industry in coating ordinary metal machine parts with a layer of metallic glass, thus imparting them with the surface characteristics of an amorphous metal. This makes the parts more durable and corrosion resistant, and also reduces friction. Attesting to their utility, one company that has taken advantage of such enhanced components, Cogentrix Energy of Charlotte, North Carolina, formerly had to replace the economizer tubes on their coal-fired boilers every six months due to corrosion. The company had their tube interiors spray-coated with a layer of amorphous metal in 1992, and as of 2004 they have yet to need replacing.
Golf clubs containing a metallic glass composite have been on the market since 1998. They are sold under the brand name Liquidmetal, a Lake Forest, California company started by Johnson's research team at Caltech. The enhanced elasticity of amorphous metal means that a Liquidmetal club returns essentially all of the energy of a golfer's strike to the ball and adding, it is claimed, as much as 30 yards to a pro golfer's drive.
The future
Because they are made from a glass, metallic glass blades are sharp straight from the mold, and remain sharp far longer than ordinary metal blades. According to Johnson, a metallic glass razor would stay sharp for an entire year, and the resulting infrequent need to replace such items is perhaps the reason why they have yet to be commercialized for the general public. However, metallic glass blades for surgical applications are in development.
Many other uses for metallic glass are expected in the near future, including artificial hips and knees, ultralight cases for laptops and cellular phones, and even military applications such as armor-piercing bullets. And with the increased strength of metallic glass alloys and the fatigue resistance of partially-crystalline composites, combined with the recent development of amorphous stainless steel, it is likely that larger structures will one day be built from metallic glass, including automobiles, bridges and even entire buildings.
Marrying the advantages of glass and metal, metallic glasses have a lot to offer our technological society. Advances continue to be made as the relations between composition, structure, and properties are further elucidated with ongoing research, particularly in the field of dynamic computer modeling to predict the results of various alloy compositions. With its unique properties and increasing ease of manufacture, metallic glass is poised to become one of the great materials of the not-too-distant future.
References
- "Amorphous metal" at Wikipedia
- "Grain boundary" at Wikipedia
- "Metallic glass: A drop of the hard stuff" at New Scientist