Nucleation, droplets, and the physics of clouds
{{#ev:youtube|g4kBNBEJKD8}} A more dangerous application of nucleation!
Nucleation is the process which results in the formation of a particle (or nuclei) of a different thermodynamic phase from a solution, liquid, or vapor. This process is very important, as almost every first-order phase transition proceeds from nucleation. Some second order processes, such as ferromagnetic reorientation and crystallization, are also caused by nucleation. There will be some basic equations and definitions for second-order nucleation, but since the physics of clouds deals with first order phase transitions (namely condensation), most of this project will be devoted to nucleation in first order phase transitions. This process occurs all throughout science and nature and has several applications, both harmless and dangerous, from the delicate formation of clouds to the explosion caused by placing Mentos in Coke. Nucleation is also very important in the development of semiconductors. In nature this process occurs at an interface, such as the surface of a container which holds the fluid or the surface of a particle of dust which is in the solution. This is referred to as heterogeneous nucleation and happens much more often than homogeneous nucleation, which occurs in completely pure liquids. This type of nucleation requires activation by supercooling or superheating the fluid. If this nucleus is the appropriate size, after nucleation the nucleus (or droplet in clouds) will continue to grow. This process is referred to as nucleation and growth, which is very important when describing precipitation in clouds.
Phase Transitions
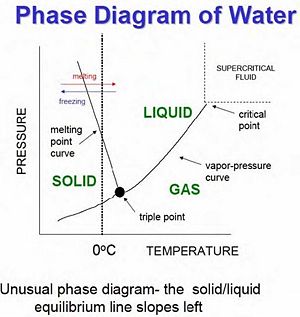
Before defining nucleation in more detail, it is necessary to start by stating some basic principles in thermodynamics and phase transitions. There are two orders of phase transitions. First order transitions refer to discontinuous jumps in phase transitions, such as condensation and freezing. Second-order transitions are more complicated and are described as continuous transitions. Ferromagnets, superconductors, and supercritical fluids all result from second order phase transitions.
Also, there are two states of matter, that which is in equilibrium and that which is not. The second law of thermodynamics tells us that the entropy of an isolated system not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium. This explains why matter self-assembles, or re-arranges itself in the most energy efficient form. After a phase transition one can find a system which is in non-equilibrium, and it will immediately begin to re-assemble. Phase transitions always change the entropy of a system, usually from a more ordered state to a less ordered state (such as melting and evaporation). In any system the free energy is the difference of the internal energy and the entropy, that is, E = U - TS, where T is the temperature of the system. Phase separation occurs when the same matter exists in two different phases in the same system (same temperature, pressure, etc.). One important difference between phase separation and nucleation is that nucleation can occur during second-order phase transitions, but phases will not separate during a continuous transition of states. In relation to the free energy there are now three types of stability that should be mentioned, which are based on the free energy:
Stable - A system in which a phase separation would cause an increase in the free energy. This phase separation doesn't happen naturally, so the system is stable and doesn't change.
Unstable - A system in which a phase separation causes a decrease in the free energy. This process will happen at any small fluctuation in the system, so the system is very unstable and usually changes immediately.
Metastable - The system is stable with respect to small fluctuations in the matter, but will phase separate with larger fluctuations. This system has an energy barrier which needs to be overcome in order for these phases to separate.
Co-existence occurs when different phase of matter exist at the same time, and a graph of the phase states as a function of temperature and pressure is referred to as the coexistence dome. The concepts of phase separation and coexistence are very important for the physics of clouds as well as other forms of matter, such as gasoline.
Nucleation Definition & Derivation
It is in this metastable state where nucleation occurs. For phase separation to occur, a drop of the material at one phase must be nucleated, using thermal activation, into a drop of the other phase. There are two factors which contribute to the formation of a droplet. An interface must be formed, which will contribute positively to the energy, and a negative contribution from the volume of the droplet. This is because the phase separation that is about to occur will decrease the free energy (the system is increasing in stability). For a system that exists over a long period of time (), the energy E that is required to form of a droplet of liquid matter can be defined as:
,
where is the surface tension, S(t) is the surface area with respect to time, h is a small chemical potential, and V(t)is the volume with respect to time. The sphere is geometrically the most efficient shape for the droplet to be in be in because it minimizes the area of interface between the two drops, and in nature this is the shape that the droplet takes. Substituting the surface area and volume of a sphere into the equation, we get:
Taking the derivative and setting it equal to zero, we can find the maximum (critical) droplet size:
Therefore, the radius of the critical droplet is dependent on the surface tension of the droplet and the chemical potential of the material. If the droplet will grow (nucleation and growth), and if the droplet will shrink. The probability of any particle nucleating is proportional to the Boltzmann factor, that is:
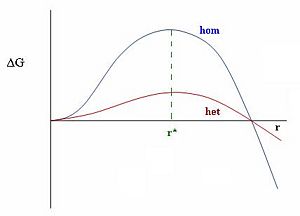
Looking at the graph on the left one can see the large energy difference between heterogeneous and homogeneous nucleation. A state without any impurities requires a lot of energy to overcome the energy barrier before nucleation. This helps to explain how we can have "supercooled" water in the atmosphere, tiny water droplets which can be much colder than their usual freezing point of zero degrees Celsius. Adding tiny particles, such as dust or "seeds" for inducing cloud precipitation, lowers the surface tension required by the droplet because it can use these particles as surfaces with which to nucleate. As the surface tension decreases, both the critical radius of the droplet and the energy required to form it decrease.
Application: The Physics of Clouds
Our increased knowledge of phase transitions and nucleation have led to a much better understanding of meteorology and weather patterns, especially the formation of clouds. These clouds are formed by condensation which is triggered by nucleation. One application of nucleation in weather is referred to as cloud seeding. We can even alter the times at which it rains by seeding clouds with cloud condensation nuclei.
Cloud Formation & Precipitation
Clouds are formed when water vapor which is brought into the upper atmosphere by updraught condenses and droplets form. When enough of these droplets cluster together, we can see clouds. Here nucleation is important because these particles will not nucleate below supersaturation without cloud condensation nuclei, or aerosols, with which the water vapor particles can nucleate. In the atmosphere a particle with roughly the size of 0.1 micrometer can nucleate a cloud droplet when the relative humidity is about 101% (barely supersaturated). One important atmospheric aerosol is regular table salt, NaCl, because it can absorb water vapor and cause nucleation and growth below the saturation point (100% relative humidity). Atmospheric aerosols can vary greatly in size, from 1 nanometer to around the order 100 micrometers. Aerosols in general will reflect light back into the atmosphere; they often appear white to us because clouds reflect most of the visible light (70%-95%) that they come into contact with. However, some aerosols with absorption properties will actually absorb radiation from the sun, which will heat up the atmosphere. This, when combined with the "greenhouse gas" emitted from cars and factories, are both important factors in the concept of global warming, although this effect has not really been quantified yet. Most clouds are found in the lowest section of the atmosphere, from 0 to 8 kilometers, but some clouds have been observed as high as 85 km off the surface of the Earth. While there are several different classes and types of clouds, they are all formed by the same basic process of nucleation and growth.
Precipitation occurs when these small cloud particles collide and combine (also referred to as coalescence) to become big enough and heavy enough that the air currents in the atmosphere cannot hold them, and they fall out of the cloud. And for visible rain these particles must be big enough to survive the journey from the cloud to the ground. This requires several collisions, as raindrops are about 100 times larger than cloud droplets, which are around the order of 10 micrometers. This means that there are around 1 million cloud droplets in one raindrop!
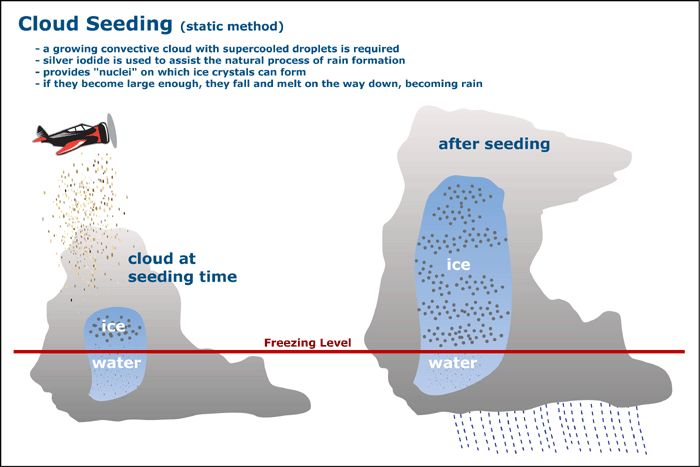
Cloud Seeding
Another way that precipitation occurs is through the process of cloud seeding, in which one can nucleate ice crystals from the cloud droplets by placing freezing nuclei inside the clouds. After clouds form some continue to rise into the atmosphere, with the temperature dropping below zero degrees Celsius. These small supercooled water droplets which are found in clouds can be as cold as -40 degrees Celsius (after that point they spontaneously freeze), but usually exist around -20 degrees Celsius. For clouds that are warmer and contain mostly water droplets, rain can be produced artificially by simply seeding the clouds with salt. However, when these clouds contain supercooled liquid and ice, more of the droplets must be nucleated into ice crystals by using a different nucleation seed.
Many scientists began predicting that one could produce rain by seeding clouds in the 1930's, but experiments weren't performed until 1946, when Vincent J. Shaefer of the General Electric Company dropped dry ice into a supercooled cloud, which precipitated snow which fell for about 2000 feet before evaporating back into the atmosphere. However, in that same year an important discovery was made by B. Vonnegut, who found that silver iodide atoms served as very efficient ice nuclei. Several experiments have been performed since then using a cloud chamber which have shown that silver iodide and lead iodide are the best seeds for artificial precipitation. This is because both molecules have hexagonal symmetry similar to ice, and also because they are insoluble in water. Silver iodide is still the most commonly used seed, and it is distributed to the clouds through smoke. For small systems it is usually distributed by a plane, but for larger systems smoke is created from the ground and carried into the upper atmosphere by the associated updraught.
While the experimental results of how much rain is formed is still uncertain, the concept of cloud seeding has been used in several instances, most notably in the recent 2008 summer Olympics, when the Chinese government seeded the clouds to clear out pollution with the rain and also to keep it from raining during the opening ceremony. Cloud seeding and the quest to control the weather and precipitation should continue to be a heavily researched topic for years to come.
Sources
Soft Condensed Matter, by Richard A. L. Jones (Oxford University Press, 2007).
Encyclopedia of Applied Physics, edited by George L. Trigg (VCH Publishers, Inc., 1991), Volume 1
Encyclopaedic Dictionary of Physics, edited by J. Thewlis (Pergamon Press, 1961)
Encyclopedia of Mathematical Physics (Elsevier, 2006), Volume 3