Phase transitions in biological systems
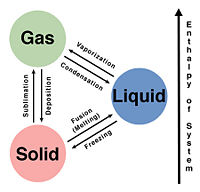
Phase transitions are present everywhere. Even within something as complex as the human body or plants, phase transitions are still present throughout processes which these systems use to survive. Critical phenomena such as second-order phase transitions are hard to find in certain biological systems however first-order phase transitions are easily present. Trying to analyze a biological system to define that phase transitions that occur within the system can sometimes be difficult but they are there. Phase transitions can be defined as a change from one phase to another, for example water boiling from a liquid phase into a gaseous phase. These phase transitions allow matter to exist in various forms such as solids, liquids, and gases. There are several other forms of matter, however for simplicity in biological systems only these three will be relevant. Each of these phases exhibit properties which allow differentiation between one another such as density and volume. There are two kinds of phase transitions; first order and second order. First order phase transitions can be described thermodynamically by having a latent heat, which is energy that is required by a substance in order to go from one phase to another. In a biological system, these will be more prevalent than second order phase transitions. Second order phase transitions do not have a specific latent heat and usually occur under specific conditions such as increasing or decreasing temperature by heating or cooling. These temperatures typically are not capable of being reached within a biological system because these temperatures can be detrimental to the system. Several examples of second order phase transitions include superconductivity and superfluids. In a biological system such as the human body, expect to find phase transitions of the first order. Physical phase transitions such as ice melting and water freezing can usually be reversed, however in a biological system this is not always the case. The fertilization of an egg and the phases or development cannot be reversed. Much is the same with viral phase transitions.
Biological Materials as Soft Condensed Matter?
Why yes! Many biological materials can be considered to be soft condensed matter. Believe it or not, but muscles and blood exhibit properties which allows them to be classified as soft condensed matter. Blood can be classified as a colloid, which is a chemical substance consisting of a mix of various things. The various things that make up a colloid are not dissolved in it, but rather are distributive evenly within the substance. The neat thing about colloids is that they're able to exist in gaseous, liquid, and solids forms.
The Human Body as a Biological System
The human body is a very complex biological system with many organ systems within itself that allow bodily process to maintain life. Within these systems, various phase transitions do occur. Many of these phase transitions are necessary in order for things to work. Freezing typically does not occur in the human body, nor does water ever boil to become gas. What occurs instead is a series of reactions that can cause these phase transitions.
Respiratory System
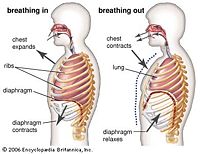
The respiratory system is an important system which allows one to exchange gases. In humans, the lungs contract by way of respiratory muscles to inhale oxygen and exhale carbon dioxide, which come in the form of gases. When a human inhales, oxygen enters the lungs and by way of diffusion, the oxygen is absorbed by the blood, which is a liquid. Diffusion is simply the transport of molecules from a high concentration to a low concentration, which usually results in a state of equilibrium. This takes place in tiny sacs that make up the lungs known as alveoli. These structures increase the surface area of the lungs which then facilitates respiration and gas exchange. Gaseous oxygen is able to be absorbed by the blood because it binds to a solid protein known as hemoglobin. This here is a phase transition because oxygen gas binds to a solid protein in liquid blood. Exhalation is a similar process. Carbon dioxide gas leaves blood by way of diffusion through alveoli and is then exhaled out of the body. As expected, these are first-order phase transitions. They play a role in the body by maintaining homeostasis and are very evident in the digestive system and metabolism.
Digestive System
For a human, digestion begins in the mouth. There are various salivary enzymes that break down solid foods such as starches. Also food is broken down by chewing, which not only makes swallowing easier, but it also increases the surface area capacity for digestion by breaking down solid particles into smaller pieces. When solid food particles enter the stomach, many acids and enzymes are released which aid in the breakdown and digestion of food. The release of a neurotransmitter known as acetylcholine, or Ach, triggers the release of more gastric juices. Protein in the stomach bonds to hydrogen ions in order to lower the pH to strong acid levels. These pH levels allow the enzyme pepsin to work optimally in order to denature proteins. Proteins can be denatured by heating them up to high temperatures however if these high temperatures were reached within a biological system, it is possible that the high temperature can cause the denaturing of proteins vital towards the survival of that biological system. This is the case with second-order phase transitions and why it is typically not found within a biological system like a human body. The denaturing process in the stomach is different because instead of using high temperatures to break polypeptide bonds in proteins, the strong acidic activity by pepsin actually reduces the structure of these polypeptide bonds. What is meant by structure is the primary, secondary, tertiary, and quaternary structure of proteins. These typically involve the sequences of amino acids that make up the protein as well as chemical bonding and atomic composition. The by-product of a lot of these reactions in the stomach is gases. Typically, acid-base reactions such as these occurring in the stomach release carbon dioxide and water as by-products so for a phase transition, we see there that the enzymatic activity of the stomach’s gastric juices in combination with solid food particles releases gas. In the stomach, liquids such as alcohols are absorbed directly into the circulatory system. They are able to pass through because they are extremely small molecules and are able to pass through the membrane of the stomach. However, the solid food does not stay solid for long. Over time, solid food begins to turn into a semi fluid mass known as chyme. Chyme is partially digested food and begins to move past the stomach into the small intestine. Once again, more digestion is occurring and nutrients begin to be absorbed by blood. What is eventually leftover is wastes and they begin to build up and move into the large intestine where water is extracted from waste and eventually expelled out of the system as a solid.
Circulatory System
The circulatory system is interesting because it works together with most of the systems in the human body. Basically, it provides the medium in which chemicals and nutrients are transported throughout the body. The circulatory system consists of many vessels that go throughout the whole human body. These vessels are known as arteries, capillaries, and veins and they carry blood. Blood is an interesting liquid because it is a colloid. Within it, it is possible to find all kinds of things. It is able to carry solid objects such as protein as well as gases such as carbon dioxide and oxygen.
Nervous System
What controls many of these systems and how messages are sent from various parts of the body is done by the nervous system. Signals are sent via neurons which are ever present throughout the body. Many events or stimuli cause changes in membrane potentials of cells. What then occurs is a series of events in which a cell can become depolarized and an action potential occurs, which generally can be from a wide variety of events from the release of hormones or neurotransmitters, to even the movement of an arm or leg. Repolarization then occurs and the cell goes back to its resting potential. The release of these hormones and neurotransmitters is interesting because they travel through the body via blood and bind to specific proteins. This is similar to what occurs in digestion when Ach triggers the release of pepsin. These are not really phase transitions by any means but they are irreversible events in a way such that the triggering of the release of pepsin to breakdown proteins cannot be undone. The proteins cannot be return back to their original state after becoming denatured.
Reproductive System
In some ways, the reproductive system shows some phase transitions. During the process of fertilization when sperm first makes contact with an egg, an initial reaction occurs where the membrane of the egg depolarizes to allow the sperm in. Once the sperm is in, another series of reactions prevents further depolarization of the egg which also prevents more sperm from entering. This is an irreversible process, or rather an irreversible phase transition. The development of the fetus and embryo can also be characterized in stages or phases, however not the same kind of phase transition such as going from solids to liquids to gases.
Homeostasis
Homeostasis is the regulation of a system in order to maintain stable and constant conditions. Within a biological system such as a human body, many factors contribute to homeostasis, many of which involve some kind of phase transitioning. Regulating temperature within the human body is an intrinsic process that involves many things. One way of cooling the human body is evaporation by means of sweating. The question is, how does sweating cool the body? During hard workouts or when in warm climates, people tend to sweat, usually resulting in some cooling sensation. What is actually happening is that the sweat is evaporating from the skin. The transition from liquid to gas of the sweat on your skin results in a loss of energy, or a net outward transfer of heat. All previous systems mentioned play some sort of role in homeostasis. Many biochemical reactions within the body also attribute to maintaining homeostasis. The body’s metabolism consists of many chemical reactions which occur in order for it to sustain life. Many metabolic pathways make up metabolism, many of which exhibit some form of phase transitioning. There are many metabolic pathways involved in the digestive system, which is a system of organs with allows the intake of food and nutrients. These are then converted into energy by digestion and then wastes are expelled. Before a human even eats, the body is stimulated in preparation for digestion by the senses such as taste and smell. This stimulation is done by means of a signaling process which can be described by the nervous system. The nervous system also allows for many phase transitions with all of its signaling mechanisms and means of getting messages across from one part of the body to another however that will be described later.
Plants as a Biological System
Photosynthesis
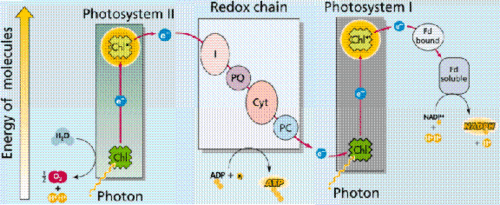
The process of photosynthesis within plants exhibit phase transitions in a big way. Plants, too, are biological systems and the process of photosynthesis is vital in their ability to survive. Photosynthesis is a process by which plants make energy by taking carbon dioxide gas and then converting it into usable, organic compounds. There are many types of photosynthesis such as C3, C4, and CAM photosynthesis. Since most plants are C3 that is what this will mostly be about. Photosynthesis is an interesting process which has many phase transitions along the way. Most plants are photoautotrophs, which mean that they are able to take sunlight, use it for energy, and create their own food from carbon dioxide gas. The transition here is from carbon dioxide gas into a solid sugar. It is from these sugars that a plant is able to survive and maintain structure. These sugars can be synthesized to become fibers in the plant as well as store energy. The process to convert carbon dioxide gas into a sugar is called carbon fixation and is supplied with energy by the process of photosynthesis. The energy that the plants, as well as humans use is called adenosine triphosphate, or ATP. So the question is, how does this process convert carbon dioxide gas into the energy required to create sugars, and in the end release oxygen gas into the atmosphere?
Photosynthesis takes place in the leaves of plants. There are pores known as stomata which allow the intake of CO2 gas and the release of O2 gas. These pores can be controlled to be open or closed. Generally, for C3 plants, they are open during the daytime and closed when the sun is down. Water is able to evaporate out of these stomata, another phase transition. Photosynthesis begins with light reactions, also known as light-dependent reactions. What happens is light excites electrons located in the chlorophyll, which raises them to a high energy state. This sparks a continuation of reactions which result in electron flow and eventually the energy becomes converted into ATP. ATP provides the energy for bonds to form. The energy flow also breaks the bonds which hold water together, thus releasing O2 gas as a by-product of photosynthesis. (Respiration is the opposite of photosynthesis in a way such that oxygen gas comes in and leaves as carbon dioxide gas.) The energy allows the CO2 gas to bond with H2O and form sugars which can then be used to create other compounds.
The Effect of Phase Transitions on Cells and Blood
Everything is made of small basic units known as cells. In biological systems such as the human body or plants, these cells are not resistant to phase transitions. Cells can be frozen and this affects them drastically. High temperatures also affect them in a harmful way, usually destroying them, or denaturing them. An example of this is cooking meats such as steak. The high temperatures denature proteins, breaking down bonds which makes the meat more tender.
The structure of cells is important when describing what occurs. The cell wall contains a membrane which is permeable to only certain things, such as lipids or fats. Since fats are affected by temperature, these membranes of these cells are affected as well. When body temperature within a human body drops, the proteins within these cells begin to clump up, and water inside and out of the cell begin to cool down and freeze. If cells are frozen in the human body or plants, they tend to die. This results from the formation of ice crystals from the freezing of water. The jagged structure of these ice crystals is what destroys the delicate membrane of the cell. This is evident when plants in the winter freeze, and die. What is actually happening is a phase transition. The human body as well as plants is made up mostly of water. When it comes to increased body temperature, the cell walls actually begin to lose their elasticity and believe it or not, can melt.
When the freezing of cells can be controlled at a rate that doesn't allow ice crystals to form, then what you have is cryogenics. Cryogenics is a technology that is able to preserve human body's by means of freezing them. The science is very interesting.
Biological Thermodynamics
Biological Thermodynamics combines the laws of thermodynamics with biology in order to explain or possibly quantify changes in energy from reactions. Many organ systems within the human body, (if not all of them), require some energy in order to function. In a plant, photosynthesis converts sunlight to energy usable by the plant so that it may synthesize sugars out of carbon dioxide gas and water. There has to be some sort of method to quantify the energy required in these reactions. The complexity of the human body results in it usually not being in equilibrium due to all of the spontaneous reactions taking place. Thermodynamics can be used to describe this.
Sources
1. “Digestion”, 15APR09, <http://en.wikipedia.org/wiki/Digestion>
2. Fox, Stuart Ira, “Human Physiology”, 10th Edition, McGraw-Hill Companies, 2008
3. “MIT Department of Physics: Biophysics: Biological and Medicine”, 15APR09,< http://web.mit.edu/physics/research/areasofresearch/atomic_cm_plasma/biophysics/biophysics.html>
4. “Nervous system”, 15APR09, <http://en.wikipedia.org/wiki/Nervous_system>
5. “Phase Changes”, 15APR09, < http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/phase.html>
6. “Phase transition”, 15APR09, <http://en.wikipedia.org/wiki/Phase_transition>
7. “Photosynthesis”, 15APR09, <http://www.emc.maricopa.edu/faculty/farabee/BIOBK/BioBookPS.html>
8. “Photosynthesis”, 15APR09, <http://en.wikipedia.org/wiki/Photosynthesis>
9. “Soft Condensed Matter: where physics meets biology”, 15APR09,< http://physicsworld.com/cws/article/print/169>
10. “Types of Photosynthesis”, <http://wc.pima.edu/~bfiero/tucsonecology/plants/plants_photosynthesis.htm>
11. "Biological Thermodynamics", <http://en.wikipedia.org/wiki/Biological_thermodynamics>
12. "Soft matter", <http://en.wikipedia.org/wiki/Soft_matter>
13. "Colloid", <http://en.wikipedia.org/wiki/Colloids>
14. "When All Hell Breaks Loose" By Cody Lundin, Russell Miller, Christopher Marchetti