Super Liquids: A Look at the Phenomena of Superfluidity and Superheated Liquids
At first glance, there doesn't seem to be much in common between Superfluids and Superheated Liquids. However, these two substances illustrate two important instances concerning symmetry breaking. By studying Superfluidity, we are looking at an example of one of the more exotic things that can happen once the symmetry of a system has been broken. We can then compare this to the phenomena of Superheated Liquids which should exhibit symmetry breaking, but which, due to various other influences, remain in a metastable state. By comparing these two intriguing phenomena, we can gain a better insight into the thought provoking world of Super Liquids.
A Brief Definition of Symmetry Breaking
Both of these phenomena arise from the occurrence of symmetry breaking. When a system starts in one phase and must transition to another, the system goes from a state where it is in equilibrium to a mixture of states. This mixture of states is the symmetry breaking.
For superfluids, the symmetry breaks and allows the particles to become Bosons, thus everything occupies the smallest Energy state.
In superheated liquids there is either a lack of nucleation or the pressure of the system is very high, thus symmetry breaking cannot occur and these liquids will occupy a metastable state.
In order for symmetry breaking to occur, a system must be sufficiently large. This means that the number of particles . Once this condition has been met, the system has reached the thermodynamic limit and spontaneous symmetry breaking can occur.
Superfluidity
What Makes a Superfluid Super?
Superfluids are fluids which have no viscosity, meaning that they have no resistance and can flow freely. This occurs because superfluids follow Bose-Einstein Statistics which means that they act as bosons and not fermions. Bosons are a type of particle which have an integer spin and they do not follow the Pauli Exclusion Principle. This is extremely significant because the Pauli Exclusion Principle states that only so many particles of a single type can exist in the same energy level. Since Superfluids disobey this principle, all of their constituent particles can occupy the same state and can even be seen to be in the same place in space. Since all of the particles can exist in the same state, all of the particles tend to go to the lowest energy state, so every particle behaves identically to every other particle in the superfluid.
Three of the most well known superfluids are Helium-4, Helium-3, and Hydrogen. It should be noted that Superfluidity is a quantum effect and that the smaller something is, the more it behaves in a quantum mechanical manner. Since Helium and Hydrogen are both extremely small, they can be seen to exhibit quantum mechanical behavior.
The Nobel Prize for Superfluidity
There is controversy over who discovered the superfluid state first, but it can be agreed that Peter Kapitza, Jack Allen and Don Misener were the first three to publish their findings in the January 1938 edition of Nature. Jack Allen and Don Misener, who were both Canadian, worked out of the Royal Society Mond Laboratory at the University of Cambridge in the UK. Kapitza worked in Moscow at the Institute for Physical Problems.[11]
However, it was Lev Davidovich Landau, a Soviet scientist and associate of Kapitza's, who was awarded the Nobel Prize in 1962 for his theories regarding superfluidity. He graduated in 1927 from Leningrad State University, now St. Petersburg State University, and within two years he traveled to Copenhagen to study at Niels Bohr's Institute for Theoretical Physics. After his studies there, he returned to his home country and eventually made his way to Moscow to the Theory Division of the S.I. Vavilov Institute of Physical Problems. This is where his work on superfluidity took place.
Super Powers of Superfluids
As mentioned earlier, superfluids act as bosons. This property is what allows superfluids to do all sorts of things which a normal liquid couldn't do.
{{#ev:youtube|2Z6UJbwxBZI}} [[1]]
As shown in the video, superfluid Helium can leak through a container. This is because there is no viscosity and no resistance when the fluid tries to pass through the microscopic pores in the container which holds it.
Another odd property is that superfluids seem to defy gravity. Superfluids naturally seek their lowest energy state, and since there is no viscosity, a superfluid suspended in a vessel will climb the walls of a vessel and actually flow out of it making the potential energy lower.
The Math Behind Superfluidity
We begin with this equation:
We can use this to calculate the number density of the Superfluid. Since where is the number of particles in the system and is the colume of the system, we can then say
When
We then get
which, when the chemical potential is less than or equal to 2.315.
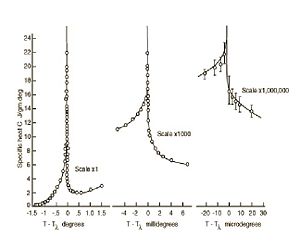
The actual temperature at which Helium-4 becomes a superfluid is 2.17 which is as predicted, below our calculated 2.315. This temperature is called the Lambda Point since the graph of the function looks like the Greek letter Lambda. The Lambda Point for Helium-4 is shown in the figure at right.
Family Ties: Other Related Phenomena
Superfluidity has been seen in Helium-3, Helium-4, and Hydrogen, but it has also been seen in gasses such as Lithium-6 [3]. Superfluidity is the liquid equivalent to the phenomenon in metals called superconductivity where there is no electrical resistance. [9]
In 1971, a paper was submitted by David Dahl and M. R. Moldover which described the "first measurements of the specific heat of a liquid in states which are metastable with respect to formation of bubbles of vapor." This is actually a description of Helium-3, not in its superfluid form, but in another state of matter which is called a superheated liquid. Since helium boils at 4.22 K, which means that is also the highest temperature at which Helium will be liquid, the temperature where this metastable state occurs is close to that of the lambda point where superfluidity occurs.
And I now believe that it is time to explore this highly interesting idea of a superheated liquid in further detail.
Superheated Liquids
A superheated liquid is a liquid in which exists in a metastable state. In this case, the internal temperature of the liquid is greater than its boiling point, but it does not boil.
All the Phases Around You
In the course of our day, we see nearly every phase of matter imaginable. We see the clouds outside which are formed from the condensation of water vapor, we see the solid desks we use for our work, and the warm liquid tea that keeps us awake. Often we see substances which defy common classification. The glass in your windows, for instance, is technically classified as an amorphous solid since glass has a very high viscosity and over time it will actually flow.
With evidence of various phase transitions around you, would you believe that even your cup of tea has the capacity to defy the normal classification of things?
A Faulty Phase Transition
Phase transitions occur when the symmetry of a system is broken. But what happens when that phase transition is suppressed? This is precisely what causes fluids to superheat.
Suppose you boil tap water in a microwave. This water has impurities in it which can help to start the water boiling by assisting in the process of nucleation. Nucleation is the process where bubbles form and it can be caused in two major ways, the first of which is an uneven heating of the liquid's surface and the second is a "roughness" either in the boiling vessel itself or in the liquid.
Since in this thought experiment we are using a microwave, we can assume that the heating of the liquid is consistant enough not to cause nucleation and we can also say that we don't have a tea bag or anything in the boiling water, since this would also induce nucleation.
Thus if the water you are boiling is pure-- perhaps you used bottled water, or maybe it was forgotten and reheated-- and the vessel you are boiling it in is particularly smooth, nucleation fails to occur and it may look as though the water hasn't boiled at all. However the instant that an impurity or "roughness" such as a teabag is introduced, the system will undergo a spontaneous phase transition and the water will "flash boil" all at once.
This is a fairly common phenomenon, one which even the Mythbusters Adam and Jamie have investigated and tested for themselves.
{{#ev:youtube|1_OXM4mr_i0}} [[2]]
Undersea Volcanoes: Mother Nature's Microwave
This phenomenon of superheating is not simply something that has been manipulated into existence through experiment. Superheated liquids can be found occurring in nature as well.
Deep in the ocean the tectonic plates which move and shape our earth allow for undersea volcanoes and vents to form. These vents occur where hot magma from the earth's mantle seeps up through the crust, either in "hot spots" which are thin spots in the tectonic plates, or at the plate boundaries. Ocean water can then seep down into the crust, and where the magma meets the water, hydrothermal vents can be formed. Most of these vents can be found along Mid-Ocean Ridges, where the plates are diverging to create new crust.
The geothermal heat from the vents causes the temperature of the water to rise well above boiling point, but due to the immense pressure at the bottom of the ocean, some 2 or 3 thousand meters down at many atmospheres, the water cannot boil. This water is actually so hot that it can melt metal.
The video below explains in great detail how these hydrothermal vents are formed. From the Woods Hole Oceanic Institute. {{#ev:youtube|rFHtVRKoaUM}} [[3]]
There are many strange things about this superheated environment. One of the strangest by far is the fact that life can be found there. Some of these vents reach temperatures of K, and yet at above the boiling point of ocean water at sea level, these organisms, some of them as complex as crabs and snails, can thrive.
This video is from a BBC documentary on undersea hydrothermal vents.
{{#ev:youtube|4LoiInUoRMQ}} [[4]]
Practical Applications of Superheated Liqids
Through the study of these hydrothermal vents which have superheated seawater, many new applications and technologies have been discovered.
Hydrothermal vents fume "smoke" which is really a mineral rich liquid. Those which spew black "smoke" typically contain copper, iron, and zinc oxides. These are minerals which are being mined on land, and finding these vents has opened up another area to gather resources. It has also been determined that the land deposits which are currently being mined originated in the ocean. This has been proven by looking at the composition of the surrounding rocks.
Even the Human Genome Project has benefited from the study of this superheated environment, since microorganisms which live around the vents were used to help in the genome sequencing.
Apart from the hydrothermal vents, laboratory experiments with superheated liquids have also led to new uses for the superheated liquids themselves. Superheated droplets are being used in dark matter detectors because they are insensitive to gamma rays of energy under 6MeV. The idea is that as a particle, hopefully a WIMP (Weakly Interacting Massive Particle), hits the superheated liquid it will give it just enough energy to cause nucleation. This can then be detected and recorded. These types of detectors are safer than their predecessors because this type of detector has many small drops of superheated liquid instead of a large container. This minimizes the danger of spontaneous boiling which could cause injuries.[9]
Conclusion
It is only through the persistant study of strange phenomena that we can continue to advance the realms of science and knowledge. Perhaps one day someone will find a superfluid which is close to room temperature which can then be used for new technologies. Perhaps the organisms that are found in the superheated waters surrounding the hydrothermal vents will be found to hold the key to curing diseases like cancer or AIDS. Strange and wonderful phenomena surround us, and it is up to us as scientists to bring attention and understanding to these incredible marvels.
References
- [Link to an Introduction of Superfluidity]
- [The Russian Academy of Science's Evidence for Hydrogen superfluidity(2000)]
- [MIT's 2005 high temperature superfluid gas of Lithium]
- [|Metastable Thermodynamic States Near the Critical Point of 1971]
- [|Experimental Properties of Superfluid ]
- [Life]
- [| Physical Review B 73, 012201 (2006)]
- [| Physical Review E Vol. 62, 4 Oct. 2000 "How High Can the Temperature of a Liquid be Raised Without Boiling?"]
- [| Physical Review D Vol. 54, 2 July 1996 "Superheated Microdrops as Cold Dark Matter Detectors"]
- [| Superfluidity and Quantized Vorticies]
- ["Superfluidity: three people, two papers, one prize"]