PHZ3400 Phase Transition: Difference between revisions
| Line 17: | Line 17: | ||
==Why Ice Floats? Consequences.== | ==Why Ice Floats? Consequences.== | ||
An object will float if it is less dense than the fluid that it has been placed in. For an object of mass m to float, it must displace mass m of liquid. | |||
Water reaches its maximum density at 4 degrees C and then continues to freeze. When it freezes, the solid ice is less dense than the liquid water. Usually, the liquid state of a material is less dense than the solid. The difference in the case of water is hydrogen boning. | |||
The water molecule is composed of two hydrogen atoms and one oxygen atom held together by covalent bonds. The molecules have a weak attraction to each other in the form of a hydrogen bond. This occurs between the positively charged hydrogen atoms in the molecule and the negatively charged oxygen atoms in another molecule. When water freezes into a solid, the hydrogen bonds adjust such that the negatively oxygen atom in each molecule are far apart. This creates a lattice that is less dense than the liquid form. | |||
==Van der Waals Equation== | ==Van der Waals Equation== | ||
[[Image:Johannes_Diderik_van_der_Waals.jpg|thumb|right|200px|Johannes Diderik van der Waals]] | [[Image:Johannes_Diderik_van_der_Waals.jpg|thumb|right|200px|Johannes Diderik van der Waals]] | ||
Revision as of 15:56, 29 January 2009
Phases of Matter
Matter can exist in many phases. The phases most common are solids, liquids, and gases. In Physics, a phase can be described as a region of space in which all physical properties of a material remain constant, or uniform. Having consistent physical properties and chemical uniformity allow one to distinguish between the various phases, or states of matter.
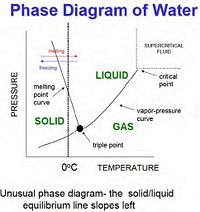
Gas-Liquid-Solid Phase Diagram
When studying phase transitions, it is important to be able to read and understand a phase diagram. A phase diagram is a pressure vs. temperature graph of an element or compound that shows which phase we can expect it to be in (see right for example). The lines represent the boundaries between phases, and at any point on these lines we can expect two (or even three) phases to exist simultaneously in equilibrium. In the area between curves only one phase of matter exist. There are two important points on phase diagrams which should be mentioned:
Triple Point - At this specific temperature and corresponding pressure, the three phases of matter can exist simultaneously in equilibrium. This means that evaporation, condensation, freezing, melting, fusion, and sublimation are happening at once.
Critical Point - At this point in temperature and pressure (and at any point above this), the boundary between liquid and gas doesn't exist any more. Here a gas cannot be distinguished from a liquid, and the heat of vaporization is zero. However, you can still have a smooth transition between gas and liquid by going up and around the critical point (as seen on the phase diagram) by passing through the supercritical fluid region.
Phase Separation and Nucleation
This we'll cover on Friday, Jan. 30
Why Ice Floats? Consequences.
An object will float if it is less dense than the fluid that it has been placed in. For an object of mass m to float, it must displace mass m of liquid.
Water reaches its maximum density at 4 degrees C and then continues to freeze. When it freezes, the solid ice is less dense than the liquid water. Usually, the liquid state of a material is less dense than the solid. The difference in the case of water is hydrogen boning.
The water molecule is composed of two hydrogen atoms and one oxygen atom held together by covalent bonds. The molecules have a weak attraction to each other in the form of a hydrogen bond. This occurs between the positively charged hydrogen atoms in the molecule and the negatively charged oxygen atoms in another molecule. When water freezes into a solid, the hydrogen bonds adjust such that the negatively oxygen atom in each molecule are far apart. This creates a lattice that is less dense than the liquid form.
Van der Waals Equation
The Van der Waals Equation is an equation of state, that is a modification of the ideal gas law,
, that takes into account the non-zero size of particles as well as attractive forces (Van der Waals Force) felt between them.
Johannes van der Waals proposed two modifications to the ideal gas law:
Van der Waals Equation:
where
is pressure
is temperature
is the number of particles
is volume of the container containing the fluid
is the measure of attraction between particles
is the volume excluded by a mole of particles. We know that each atom or a molecule in a liquid has a very steep repulsive potential at short distances, due to the Pauli exclusion principle. Thus a volume comparable to the atomic radius is essentially excluded to other particles. This considerably limits the volume of space accessible to thermal motion of every atom or molecule, an effect that van der Waals included phenomenologically in his theory. Today we have more accurate microscopic theories that accurately computed the effects associated with liquid correlations, and provide a microscopic basis for the van der Waals equaton.